RESEARCH IN A NUTSHELL
Research is complicated and difficult for non-scientists to understand? Wrong!
We want to make our students' research clear.
Whether thesis, research project or publication - we explain it in three sentences:
1. What is it basically about
2. What do we do in concrete terms
3. What is our goal
Find here - understandable and „in a nutshell“ - what our students are researching.
Meshal Ansari, PhD Student (Schiller Lab, LHI)
T cells are essential in human immune response and clearance of virus-infected cells. We aimed to capture T cells that react specifically to SARS-CoV-2 after manually stimulating them with parts of the virus.
We could show the correspondence of our artificially activated T cells to those from the respiratory tract of severely diseased COVID-19 patients.
This work characterizes the consequences of a viral infection on a cellular level and demonstrates how understanding T cell reactions can be leveraged to guide therapies and vaccines for (emerging pathogens such as) SARS-CoV-2.

CT showing pneumonia, caused by Covid-19 (right) - © LMU clinic Munich
Find the publication here:
Single-cell RNA sequencing reveals ex vivo signatures of SARS-CoV-2-reactive T cells through 'reverse phenotyping
David S. Fischer et al, Nature Communications 2021
Lin Yang, Postdoctoral Fellow (Schmid Lab, LHI)
Project/Publication: Three-Dimensional Quantitative Co-Mapping of Pulmonary Morphology and Nanoparticle Distribution with Cellular Resolution in Nondissected Murine Lungs
Depending on the origin of lung diseases occured, pulmonary drugs should be delivered to specific regions such as the lung airways for asthma and COPD and lung alveolar sacs for lung cancer.
To understand and visualize the distribution and biological fate of drug aerosols delivered via nanoparticles into murine lungs, we used complementary imaging methods.
The findings provide fundamental knowledge and guidance for the development of new inhalation methods and aerosol drugs with a final goal for improved inhalation therapy.

Nanoparticles (red) in murine lung tissue (green)
Find the publication here:
Three-Dimensional Quantitative Co-Mapping of Pulmonary Morphology and Nanoparticle Distribution with Cellular Resolution in Nondissected Murine Lungs
Lin Yang et al., ACS nano 2019
Christoph Mayr, Postdoctoral Fellow (Schiller Lab, LHI)
Project/Publication: Integrative analysis of cell state changes in lungfibrosis with peripheral protein biomarkers
Future personalized interceptive medicine wants to assess early onset of disease in a pre‐clinical stage when the patient is still feeling healthy. To work toward this goal, we must understand the changes to cellular circuits in patient organs early in disease.
We showed that data transfer between single‐cell transcriptomic and body fluid proteomic modalities is possible and identified specific gene expression and frequency changes on cell type level and used histopathologically staged data to infer early changes in lung fibrosis patients.
The work provides an integrated single‐cell atlas of human lung fibrosis and novel biomarker signatures for monitoring the disease progression in lung fibrosis.

Find the publication here:
Christoph Mayr et al., EMBO 2021: Integrative analysis of cell state changes in lungfibrosis with peripheral protein biomarkers
Ali Doryab, PhD Student (Schmid Lab, LHI)
To find new therapies for chronic respiratory diseases, researchers in their in vitro experiments have to imitate the complex environment and physiology of the human lung, which is very difficult.
For these experiments, we developed a membrane that mimics the main characteristics of the basement membrane in the lung alveolar.
This membrane offers considerable advantages over currently used membranes in terms of supporting cell growth and following cell-stretch experiments that may lead to more biomimetic in vitro models for translation of in vitro response studies into clinical outcome.
Find the whole publication here:
A Biomimetic, Copolymeric Membrane for Cell‐Stretch Experiments with Pulmonary Epithelial Cells at the Air‐Liquid Interface
Doryab A. et al., Advanced Functional Materials, 2020
Maria Camila Melo Narvaez, PhD Student (Lehmann Lab, LHI)
Most chronic and acute lung diseases are prevalent in the elderly population and have no cure, leaving lung transplantation as the only option.
For this reason we summarize the current knowledge about the regenerative capacity of the aged and diseased lung.
We further review the newest developments for relevant human preclinical models that are important for the development of potential therapeutic applications.

Find the whole publication here:
"Lung regeneration: implications of the diseased niche and ageing"
M. Camila Melo-Narváez et al., European Respiratory Review, 2020
Pushkar Ramesh, PhD Student (Rinkevich Lab)
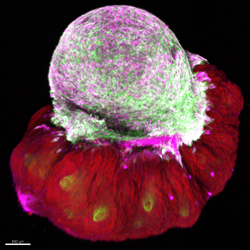
Large deep wounds in mammals caused due to surgical or traumatic wounds resolves with exuberant scars whereby the entire process of scar formation and the cellular mechanisms are incompletely understood.
In our study, we establish a simple tissue culture based assay termed “Scar-like tissue in a dish“ to create and visualize scar development in real time and to identify the prime molecule that orchestrates this scarring process.
Our aim is to utilize this tissue culture model to identify and modulate relevant cellular pathways to eventually achieve scarless wound healing.
Find the publication here:
Injury triggers fascia fibroblast collective cell migration to drive scar formation through N-cadherin.
Jiang D et al., Nature Communications, 2020